Coffee Cup Calorimeter Lab Report

Coffee Cup Calorimetry HCl-NaOH Jennifer Duong Quinn Schmidt Sabrina Mowder ResultsDiscussion Materials.
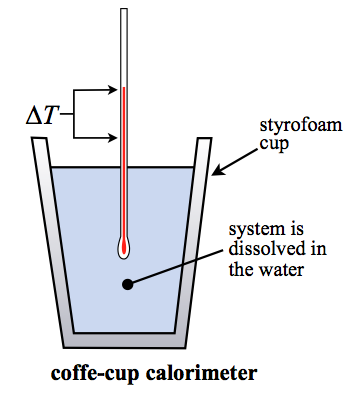
Coffee cup calorimeter lab report. Set up the coffee-cup calorimeter as shown in the previous experiment in Figure 17-1. Calorimetry Abstract The purpose of this experiment was to perform a calorimetry experiment with the use of a coffee cup calorimeter apparatus to observe the temperature change of a chemical reaction. They need to be readable and need to be provided in a clear and succinct manner.
The heat exchange between the unknown metal sample and a measured amount of water in an insulated coffee-cup calorimeter was observed and measured in order to. Styrofoam Cup Calorimeter Lab Report - Laboratory reports are utilized to describe the research study results. Save Save formal lab report 2 - calorimetry For Later.
When 100 mL of 0200 M NaClaq and 100 mL of 0200 M AgNO 3 aq both at 219 C are mixed in a coffee cup calorimeter the temperature increases to 235 C as solid AgCl forms. A bomb calorimeter works in the same manner as a coffee cup calorimeter with one big difference. Purpose In this experiment you will use a coffee-cup calorimeter to measure the temperature change.
CH 204- Intro to Chem Practice Experiment 3-Enthalpy of Chemical Reaction Dana Lucas Robin Brown TA. Coffee cup calorimeter Hot plate 100 mL Beaker Test tube holder In-Lab Experimental Procedure. The metal sample will be heated to a high temperature 100oC then placed into a coffee cup calorimeter containing a known amount of water.
University of Connecticut Stamford Campus CHEM 1127 Laboratory Fall 2020 0nline course Experiment 10 Constant-Pressure Calorimetry Instructions. Lewiss Medical-Surgical Nursing Diane Brown Helen Edwards Lesley Seaton Thomas. Heat of heat of physics 1.
Disposable pipettes 1 Conclusion Introduction Our lab results show that we understood the lab but we did not get the right values for our DHrxn and this may be because. Of hot metal to a coffee cup calorimeter. Chris February 19 2018 Introduction The purpose of this experiment was to calculate the change in enthalpy of 2 reactions using Hess Law by using a coffee calorimeter to measure the temperature changes in the sub reactions for MgO and a.