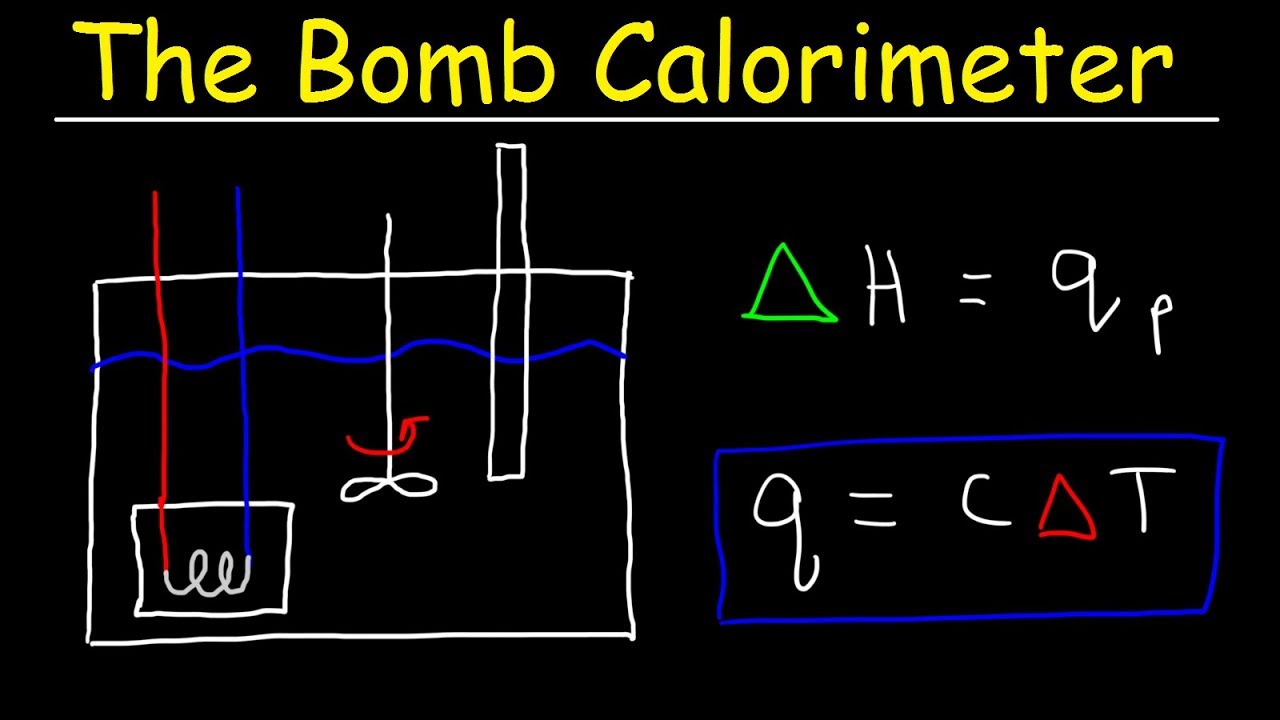
Coffee Cup Calorimeter Formula
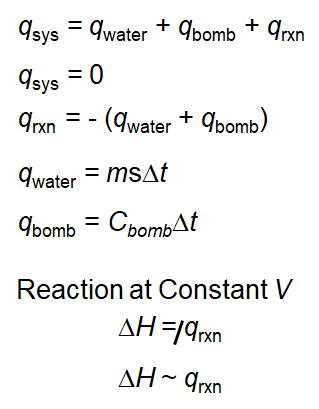
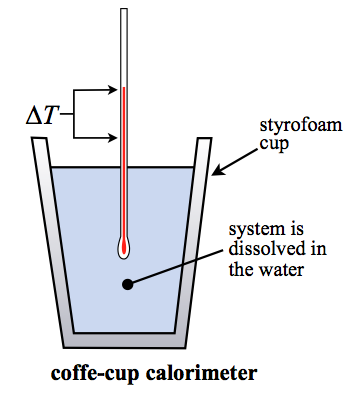
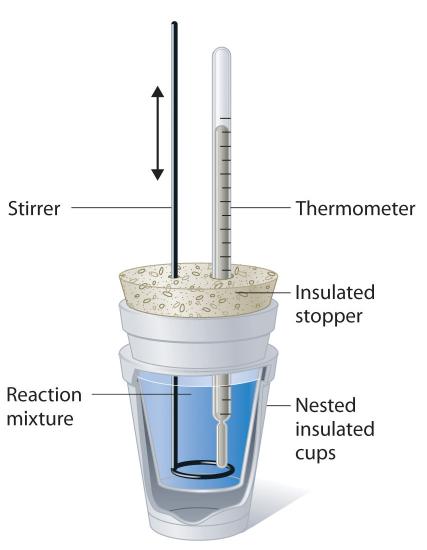
The inner cup holds a known amount of a solvent usually water that absorbs the heat from the reaction.
Coffee cup calorimeter formula. Where q is heat in joules m is mass in grams or kilograms C is specific heat and DT is change in temperature in Kelvin note. This video contains about 1. Thus Q i c e m C D T.
When 100 g of NaOHs is dissolved in 1000 mL of H 2 O l in a coffee cup calorimeter the temperature of the water rises from 2500 to 2766 oC. These easy-to-use coffee cup calorimeters allow more heat exchange with their surroundings and therefore produce less accurate energy values. A coffee cup calorimeter is typically used for solution based chemistry and as such generally involves a reaction with little or no volume change.
As such the heat that is measured in such a device is equivalent to the change in enthalpy. Since the mass of this water and its temperature change are known the value of Q calorimeter can be determined. Analysis of the heat flow is a bit more complex than it was for the coffee cup calorimeter because the heat flow into the metal parts of the calorimeter must be taken into account.
Data collected during a constant-pressure calorimetry experiment can be used to calculate the heat capacity of an unknown substance. In the laboratory we often use a simple device that is called the coffee cup calorimeter. Here the calorimeter is considered to be the water in the coffee cup.
From the balanced reaction equation. Latex-q_textmetalq_textwaterlatex Substituting in our above equation we get. A coffee cup calorimeter is a constant pressure calorimeter.
2146 k J 000341 m o l 6 293 k J m o l. Chemical reactions involve the release or consumption of energy usually in the form of heatHeat is measured in the energy units Joules J defined as 1 kgm2s2. M C DT.